|
V RaptureChrist Newsletter
|
||||||||||||||||
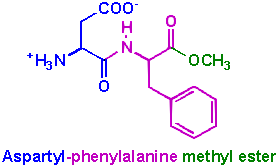
There are millions of Americans out there who believe that by drinking a Diet Coke they are doing something good for themselves. They think they are consuming less calories, less sugar, and allowing them to eat more of other things. Part of this is true, they are consuming less sugar, but what are they replacing it with? What is really in that Diet Coke or Diet Pepsi? Do you really think that what you don't know can't hurt you? Hopefully you will want to find out, because it really is poison. Trust me this concept completely floored me when I found out that the FDA approved the sale of aspartame, a poisonous substance, to millions of Americans. I myself drank some Diet Coke when it first came out, thinking I could reduce my caloric intake, but boy was I really misled! Not only weight conscious people are attracted to diet products, but also many diabetic patients use aspartame, believing it is a safe substitute for sugar. They do not know that aspartame can worsen their diabetes and unfortunately neither do their physicians. Aspartame is marketed as NutraSweet and Equal, and it is used in over 9,000 diet foods and beverages as a substitute for sugar. It is 200 times sweeter than table sugar. It was accidentally discovered in 1965 by Jim Schlatter, a chemist at G.D. Searle. Schlatter was working on a project to explore new treatments for gastric ulcers. He was synthesizing a tetrapeptide in the lab, and one of the steps in the process was to make a dipeptide intermediate, aspartyl-phenylalanine methyl ester.
While at the lab, Schlatter inadvertently spilled a small amount of the chemical on his hands. Later that morning, he licked his finger, and noticed a sweet taste. As he pondered on what it could be, he realized it must be the aspartyl-phenylalanine methyl ester he had been working with. He knew that phenylalanine and aspartic acid, two of the components in aspartame, are amino acids present in all proteins, so he thought there would be no danger in tasting the compound. It was sweet! As we can see in the diagram, the molecule of aspartame is made up of three components: two amino acids - aspartic acid and phenylalanine - and a methyl ester. When the methyl group in aspartame comes in contact with an enzyme called chymotrypsin, which is found in the small intestine, it is changed into methanol.. The methanol is then converted to formaldehyde by the enzyme alcohol dehydrogenase. Formaldehyde is a very toxic chemical. They use it to preserve cadavers. The formaldehyde is finally metabolized into formic acid (ant sting poison). People who ingest a toxic amount of methanol develop severe metabolic acidosis, and blindness. Apartame (Nutrasweet® made by Monsanto) has been associated with blindness due to the effects of methanol on the optic nerve. The Community Nutrition Institute tried in l986 to ban aspartame because people were going blind from it. According to Dr. Morgan B. Raiford, M.D., the cases of optic nerve damage due to aspartame are pathophysiologically identical to the cases of optic nerve damage due to methanol ingestion.. Other signs and symptoms of optic nerve or retinal damage due to aspartame are blurred vision, diplopia (double vision), floaters, flashes, black spots, optic neuritis, and visual field defects such as tunnel vision. Aspartame is also related to numerous health
problems such as: Alzheimer’s disease, birth defects, brain tumors,
diabetes, Gulf War syndrome, lupus, multiple sclerosis and seizures. There is a major health concern regarding the use of aspartame by people who have a rare genetic disorder called phenylketonuria. Individuals born with this disorder cannot metabolize the amino acid phenylalanine. People with phenylketonuria must therefore avoid aspartame in their diets (Stegik and Filer, 1984). The toxic effects of high levels of phenylalanine in the blood are especially observed during pregnancy. According to Dr. Louis J. Elsas, "In the developing fetus such a rise in maternal blood phenylalanine could be magnified four to six fold by the concentrative efforts of the placenta.... Thus a maternal phenylalanine of 150 uM could reach 900 uM in the developing fetal brain cells and this concentration kills such cells in tissue culture. The effect of such an increased fetal brain concentrations in vivo would probably be much more subtle and expressed as mental retardation, microcephaly, or potential birth defects." What has finally convinced most people about the harm done from ingesting aspartame is when they see dramatic improvement of such symptoms (headache, blurred vision, memory loss, paresthesias, depression) after the patients have avoided aspartame, AND the prompt predictable recurrence of these problems when the patient resumed aspartame products, either knowingly or inadvertently. The problem with aspartame regulation by the FDA is that aspartame is classified as a "food additive," and not a "drug." Adverse reactions to a substance classified as a food additive need not be reported to a federal agency, nor is continued safety monitoring required by law. There are many people who will say that they want to see more scientific data to believe that aspartame is really a poison. Listed here are some links that will give you more scientific information on Nutrasweet and Equal (the main aspartame brands). It is impossible to present all the data in one newsletter, and that is why it is important that you go to these links and read more on this topic. Links |
Saccharin  A trade name for benzoic sulphinide, saccharin
was discovered in 1879 by accident. A chemistry research assistant
Constantine Fahlberg was working on new food preservatives when he
accidentally spilled some of the compound he had synthesized on his hands.
When he returned home and ate his dinner that night, he detected the
intense sweetness of the compound. Fahberg named the compound
saccharin after the Latin saccharum which means sugar. A trade name for benzoic sulphinide, saccharin
was discovered in 1879 by accident. A chemistry research assistant
Constantine Fahlberg was working on new food preservatives when he
accidentally spilled some of the compound he had synthesized on his hands.
When he returned home and ate his dinner that night, he detected the
intense sweetness of the compound. Fahberg named the compound
saccharin after the Latin saccharum which means sugar.
The use of saccharin, under the trademark Sweet'N Low®, increased over the years as it was the only artificial sweetener known at the time. In the 1960's several studies indicated that saccharin caused bladder cancer in rats, and the FDA moved to limit its use. Saccharin use was banned in Canada in 1977. The FDA proposed a ban in 1977; however, Congress placed a moratorium on the ban to allow for more research. This moratorium was extended seven times until finally the FDA withdrew the ban in 1991 with the clause that the manufacturers needed to include a label stating that it is known to cause cancer in laboratory animals. Cyclamates In 1937 a scientist at the University of
Illinois, inadvertently placed a lit cigarette on a pile of a powder-like
substance he was experimenting with. When he returned the
cigarette to his mouth, he noticed that the powder on the cigarette tasted
sweet. Michael Sveda, the scientist in question, had serendipitously discovered
the sweet taste of cyclamates. In 1937 a scientist at the University of
Illinois, inadvertently placed a lit cigarette on a pile of a powder-like
substance he was experimenting with. When he returned the
cigarette to his mouth, he noticed that the powder on the cigarette tasted
sweet. Michael Sveda, the scientist in question, had serendipitously discovered
the sweet taste of cyclamates.
In the 1950's Abbott Laboratories in Chicago began to market cyclamates, which became the main artificial sweetener until 1969, when the FDA under Dr. Ley's direction, banned it after realizing it caused bladder tumors in laboratory animals. However, many other countries did not act on this research, and cyclamates have continued to be used as a sweetener in those countries. In 1973, Abbott asked the FDA to allow cyclamates back on the market. The FDA, after reviewing Abbott's research, indicated that Abbott had not proved to a “reasonable certainty” that cyclamate was safe for human consumption. Not one word was said about the conflict of interest in allowing Abbott to present safety data on its own product. In 1982, Abbott again petitioned the FDA to allow the sale of cyclamates, oblivious of any health risks inherent to the compound. The FDA called on its Cancer Assessment Committee (CAC) to review the data. In 1984, the CAC concluded that “the evidence on cyclamate indicated it was safe”. It is not known if members of the CAC were influenced in any way by Abbott regarding this decision. However, in 1985 there was a separate review of cyclamates by the National Academy of Sciences in which they stated that there was evidence that the use of cyclamates "may promote the growth of tumors when it combines with other carcinogens.” Abbott Laboratories is still in the process of trying to gain FDA approval for cyclamates. Acesulfame-K  Acesulfame-K (ACE-K), approved by the FDA in
1988, is another artificial sweetener. Marketed as Sunett® by Hoechst
Celanese Corp., Somerset, NJ, it is used for table-use sweeteners, chewing
gums, beverages, and dessert mixes. ACE-K has been combined
with NutraSweet in Pepsi One, and it is also used in some flavors of
Trident sugarless gum. Acesulfame-K (ACE-K), approved by the FDA in
1988, is another artificial sweetener. Marketed as Sunett® by Hoechst
Celanese Corp., Somerset, NJ, it is used for table-use sweeteners, chewing
gums, beverages, and dessert mixes. ACE-K has been combined
with NutraSweet in Pepsi One, and it is also used in some flavors of
Trident sugarless gum.
ACE-K is a white, odorless, crystalline chemical, 200 times sweeter than sucrose, but like saccharine it has a bitter aftertaste. Unlike Nutrasweet, ACE-K remains stable under high temperatures, including temperatures reached when baking. Also, unlike Nutrasweet, ACE-K does not decompose in aqueous media. This chemical has not been properly tested; the FDA based its approval on tests that were inadequate even by the FDA's own standards. But even those tests indicate that this artificial sweetener causes cancer in laboratory animals. In l987, the Center for Science in the Public Interest (CSPI) petitioned the FDA to not approve acesulfame K, but the warning was disregarded, and ACE-K continues to be used in many diet products. Sucralose  The chemical sucralose is approximately 600 times sweeter than
regular sugar (sucrose). Sucralose, manufactured by Johnson &
Johnson, sells under the trademark Splenda®. It is a derivative of refined
sugar (three -OH groups have been substituted by -Cl). The chemical sucralose is approximately 600 times sweeter than
regular sugar (sucrose). Sucralose, manufactured by Johnson &
Johnson, sells under the trademark Splenda®. It is a derivative of refined
sugar (three -OH groups have been substituted by -Cl).Sucralose has the funniest "accidental discovery" story of all the sweeteners. Tate & Lyle, a British sugar company, was looking for ways to use sucrose. In collaboration with Prof. Leslie Hough's laboratory at King's College in London, chlorinated sugars were being synthesized and tested. A foreign graduate student, Shashikant Phadnis, misunderstood a request for "testing" as a request for "tasting," leading to the discovery that many chlorinated sugars are sweet. Sucralose was approved in April 1998 as a tabletop sweetener and for use in sodas (Diet Rite), pastries, and a number of other desserts. Because we have very few studies on Sucralose, it is not possible at this time to know all the side effects that this drug will have on the human body.
Other artificial sweeteners rearing their ugly heads are: Aclame™ - Alitame made by Pfizer God warns us not to eat bad food. These artificial sweeteners might taste similar to sugar, but they are poison. Isaiah 55:2 |
|||||||||||||||